While it is not the only state that defines a critically ill patient, shock is considered by many to be the prototypical derangement of the critically ill. With our bedside treatment goals so focused on blood pressure and cardiac output, it can be easy to forget that shock at its most basic level is defined by a deficit between oxygen delivery and oxygen demand at the cellular level.
In fact, most of our interventions are actually aimed at correcting this imbalance, usually by increasing oxygen delivery. As a consequence, there has long been interest in the administration of blood transfusions as a way of augmenting oxygen delivery in the critically ill patient.
In recent years, a large volume of research has questioned the traditional practice of red blood cell transfusion to increase oxygen delivery. In fact, current literature suggests that there may be harm caused by significant transfusion reactions that do not include the more commonly associated, and dramatic reactions, such as hemolytic reactions, transfusion-associated lung injury, and direct infection from a pathogen contaminating a blood product.
This is thought to be due to the fact that transfusions contain small amounts of donor plasma, white blood cells, antibodies, and inflammatory mediators in addition to red blood cells.
A recent meta-analysis of observational studies in intensive care unit, trauma, and surgical patients found red blood cell transfusion to be an independent predictor of mortality, nosocomial infection, acute respiratory distress syndrome, and multiorgan dysfunction syndrome.
The appreciation of the potential harm from transfusions has led to the use of terms such as “allogeneic blood transfusion” or even “liquid organ 283 transplant” to highlight the potential immunomodulating effects of transfusions.
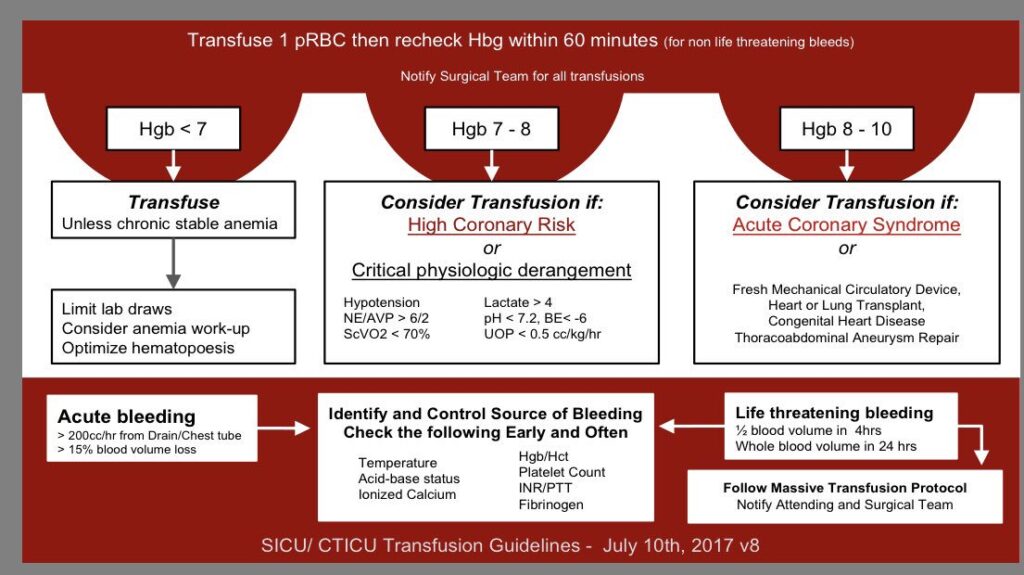
Recent studies have greatly simplified the approach to transfusion in the nonhemorrhaging critically ill patient. In the absence of acute myocardial infarction or ischemic cerebral vascular accident (CVA), critically ill patients without specific symptoms attributable to anemia should not be transfused for hemoglobin values greater than 7 g/dL.
This is best supported by randomized controlled trials comparing a hemoglobin transfusion trigger of 7 g/dL to a trigger of 10 g/dL in both a general intensive care unit population and in ICU patients with septic shock. These trials showed no benefit in using a hemoglobin transfusion trigger of 10 g/dL.
As a result of these trials and increasing evidence of an association between transfusions and adverse events, most clinicians use a hemoglobin of 7 g/dL as the transfusion trigger in nonhemorrhaging ICU critically ill patients without evidence of acute ischemia.
Patients with acute coronary syndrome (ACS) or CVA have generally been excluded from most transfusion studies, because it cannot be proved that these areas of ischemia would not resolve with an increased hemoglobin level and oxygen-carrying capacity.
Until more studies are completed, it is reasonable to follow current guidelines from the Society of Critical Care Medicine-Eastern Association for Surgery of Trauma, which state that transfusion may be beneficial in patients with an ACS and hemoglobin value less than 8 g/dL.
Key Points
- In asymptomatic, nonhemorrhaging critically ill patients, there is no specific hemoglobin threshold that requires a transfusion.
- For the majority of nonhemorrhaging critically ill patients, a hemoglobin of 7 g/dL should be used as the trigger for red blood cell transfusion.
- Some guidelines recommend transfusion in asymptomatic patients with a history of coronary disease once the hemoglobin level drops below 8 g/dL.
- The threshold for red blood cell transfusion in patients with ongoing ischemia is unclear; however, maintaining a hemoglobin level greater than 8 g/dL is reasonable.
- In the absence of hemorrhage, it is recommended to transfuse only one unit of red blood cells at a time.
Suggested Readings
- Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in highrisk patients after hip surgery. N Engl J Med. 2011;365(26):2453–2462. (https://www.nejm.org/doi/full/10.1056/nejmoa1012452)
- Hébert PC, Wells G, Blajchman MA, et al. A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. Transfusion Requirements in Critical Care Investigators, Canadian Critical Care Trials Group. N Engl J Med. 1999;340(6):409–417. https://www.nejm.org/doi/full/10.1056/nejm199902113400601
- Hebert PC, Yetisir E, MArtin C, et al. Is a low transfusion threshold safe in critically ill patients with cardiovascular diseases? Crit Care Med. 2001;29(2):227–234. https://pubmed.ncbi.nlm.nih.gov/11246298/
- Holst LB, Haase N, Wetterslev J, et al. Lower versus higher hemoglobin threshold for transfusion in septic shock. N Engl J Med. 2014;371(15):1381–1391. https://www.nejm.org/doi/full/10.1056/nejmoa1406617
- Marik PE, Corwin HL. Efficacy of red blood cell transfusion in the critically ill: A systematic review of the literature. Crit Care Med. 2008;36(9):2667–2674. https://pubmed.ncbi.nlm.nih.gov/18679112/
- Napolitano LM, Kurek S, Luchette FA, et al. Clinical practice guideline: Red blood cell transfusion in adult trauma and critical care. Crit Care Med. 2009;37(12):3124–3157. https://pubmed.ncbi.nlm.nih.gov/19773646/