Table of Contents
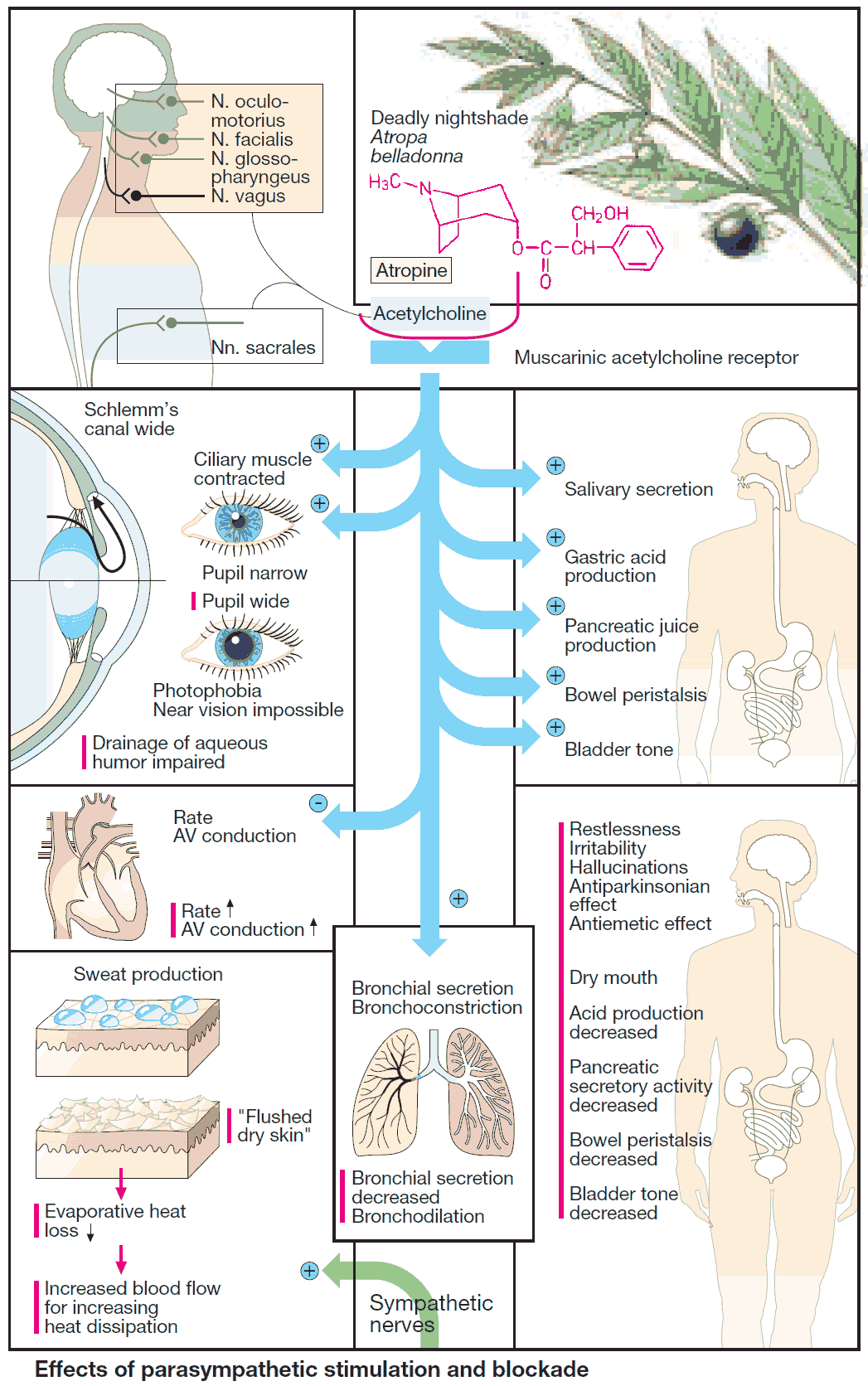
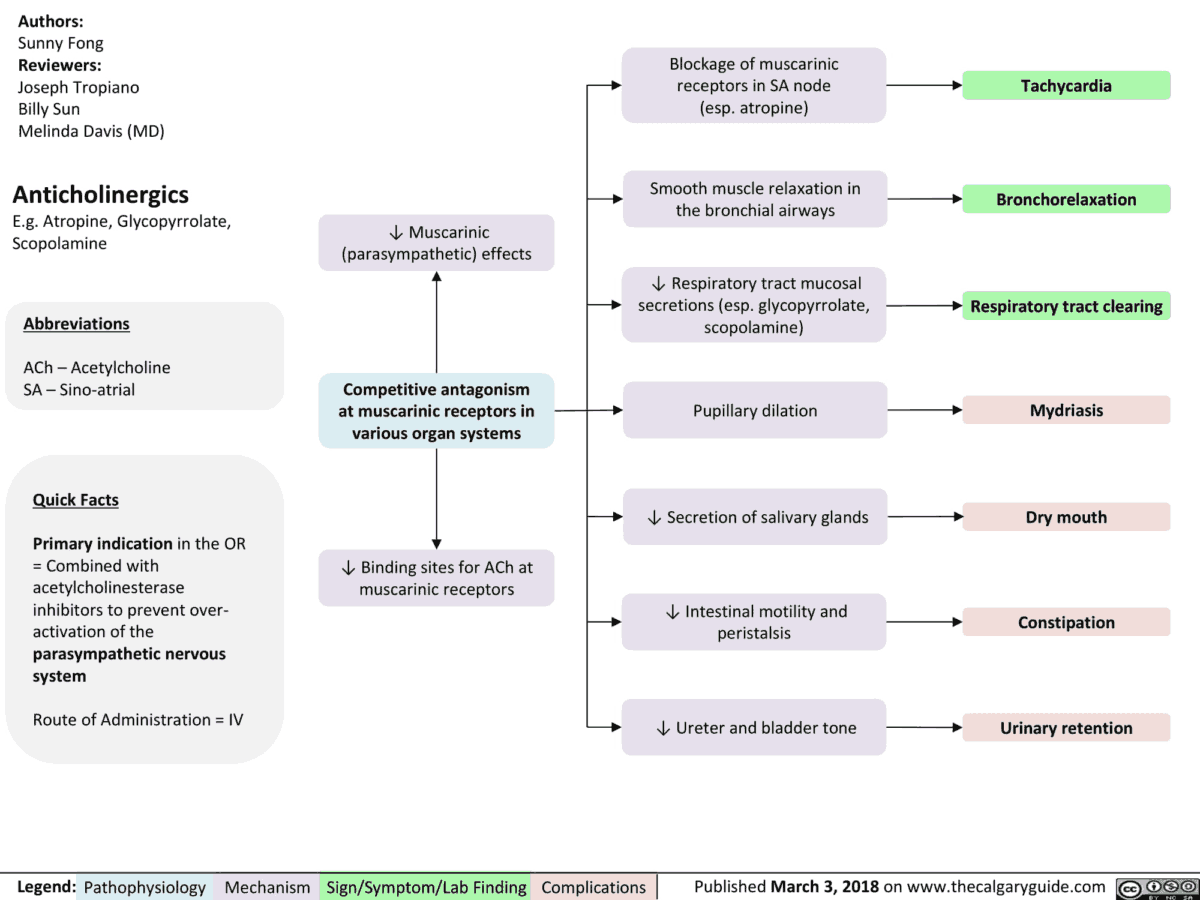
Excitation of the parasympathetic division of the autonomic nervous system causes release of acetylcholine at neuroeffector junctions in different target organs. The major effects are summarized in (image). Some of these effects have therapeutic applications, as indicated by the clinical uses of parasympathomimetics.
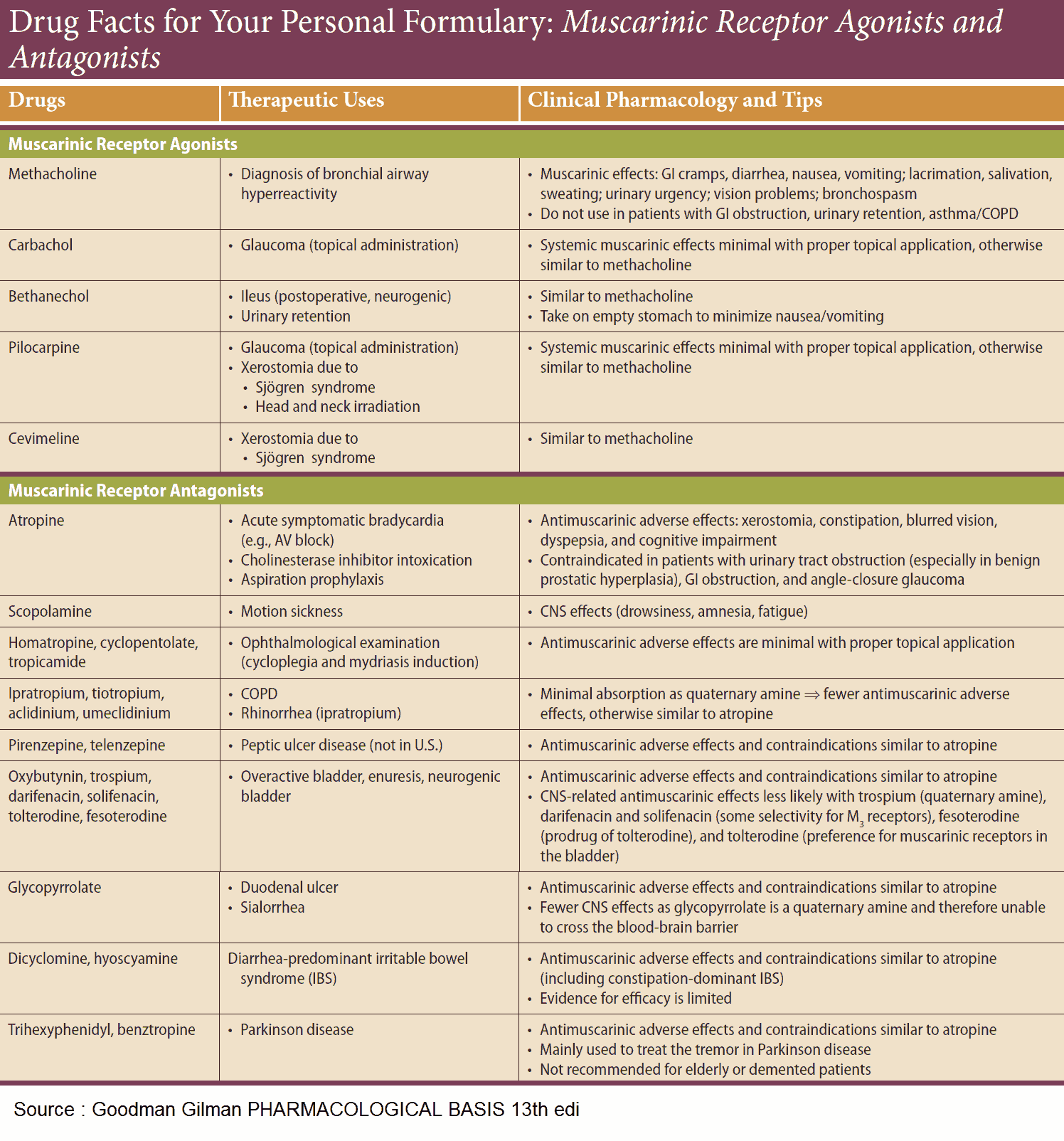
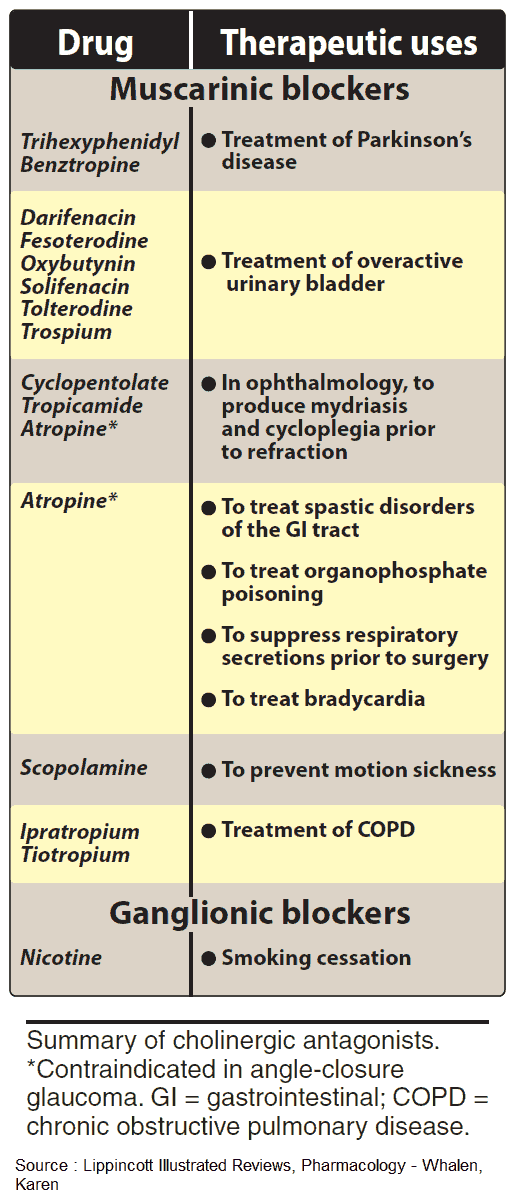
Substances acting antagonistically at the M-cholinoceptor are designated parasympatholytics (prototype: the alkaloid atropine; actions shown in red in the panels). Therapeutic use of these agents is complicated by their low organ selectivity.
Possibilities for a targeted action include:
- local application
- selection of drugs with either good or poor membrane penetrability as the situation demands
- administration of drugs possessing receptor subtype selectivity
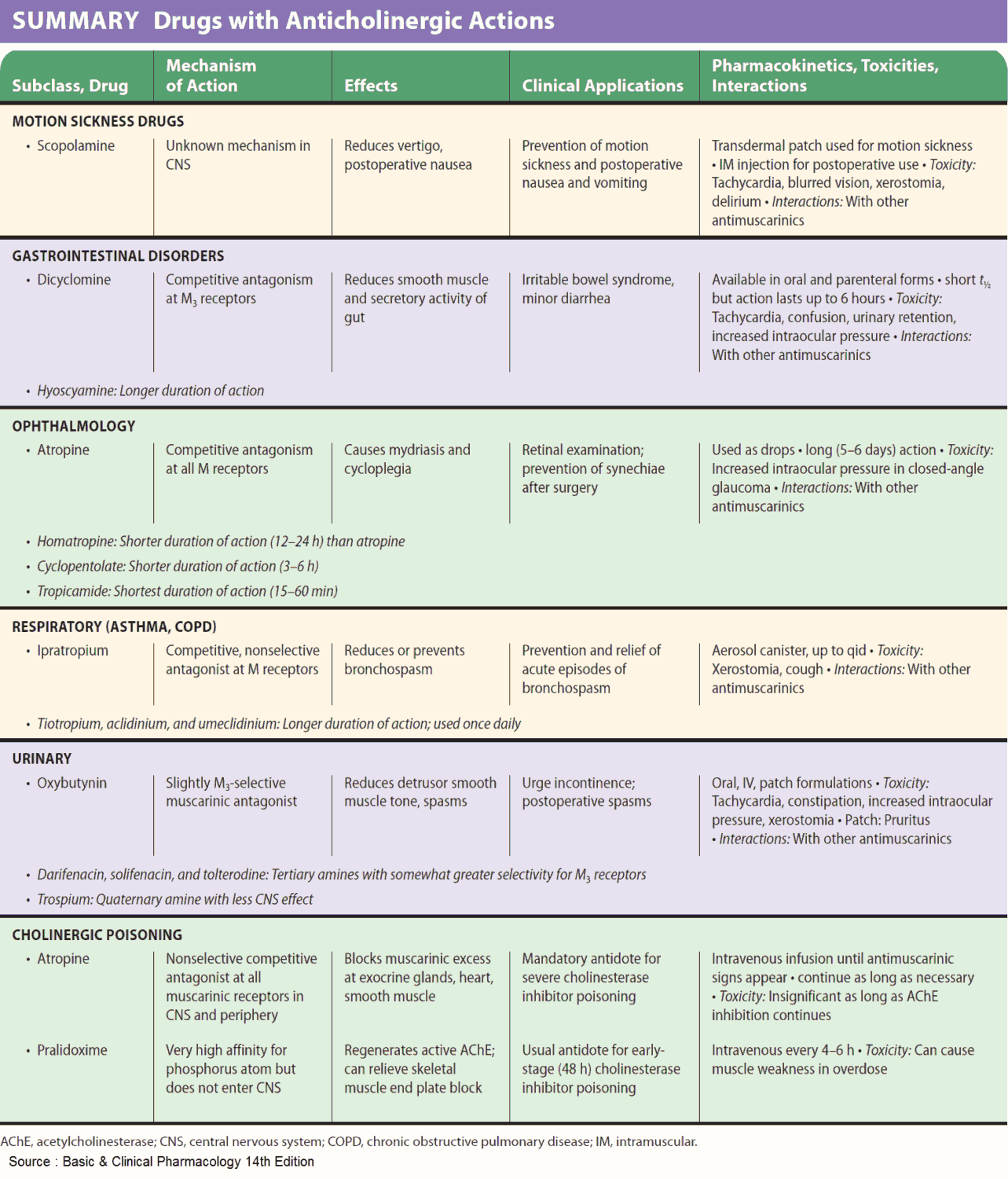
Uses and Indications of Parasympatholytics (Anticholinergics) Agents
- Inhibition of exocrine glands
- Relaxation of smooth musculature
- Cardioacceleration
- CNS-dampening effects
1. Inhibition of exocrine glands
Inhibition of Bronchial secretion
Premedication with atropine before inhalation anesthesia prevents a possible hypersecretion of bronchial mucus, which cannot be expectorated by coughing during intubation (anesthesia).
Inhibition of Gastric secretion
Stimulation of gastric acid production by vagal impulses involves an M-cholinoceptor subtype (M1-receptor), probably associated with enterochromaffin cells. Pirenzepine displays a preferential affinity for this receptor subtype. Remarkably, the HCl-secreting parietal cells possess only M3-receptors.
M1-receptors have also been demonstrated in the brain; however, these cannot be reached by pirenzepine because its lipophilicity is too low to permit penetration of the bloodbrain barrier. Pirenzepine was formerly used in the treatment of gastric and duodenal ulcers.
2. Relaxation of smooth musculature
Bronchodilation can be achieved by the use of ipratropium in conditions of increased airway resistance (chronic obstructive bronchitis, bronchial asthma). When administered by inhalation, this quaternary compound has little effect on other organs because of its low rate of systemic absorption.
Spasmolysis by N-butylscopolamine in biliary or renal colic. Because of its quaternary nitrogen, this drug does not enter the brain and requires parenteral administration. Its spasmolytic action is especially marked because of additional ganglionic blocking and direct muscle-relaxant actions.
Lowering of pupillary sphincter tonus and pupillary dilation by local administration of homatropine or tropicamide (mydriatics) allows observation of the ocular fundus. For diagnostic uses, only short-term pupillary dilation is needed. The effect of both agents subsides quickly in comparison with that of atropine (duration of several days).
3. Cardioacceleration
Ipratropium is used in bradycardia and AV-block, respectively, to raise heart rate and to facilitate cardiac impulse conduction. As a quaternary substance, it does not penetrate into the brain, which greatly reduces the risk of CNS disturbances (see below).
Relatively high oral doses are required because of an inefficient intestinal absorption. Atropine may be given to prevent cardiac arrest resulting from vagal reflex activation, incident to anesthetic induction, gastric lavage, or endoscopic procedures.
4. CNS-dampening effects
Scopolamine is effective in the prophylaxis of kinetosis (motion sickness, sea sickness); it is well absorbed transcutaneously. Scopolamine (pKa = 7.2) penetrates the blood-brain barrier faster than does atropine (pKa = 9), because at physiologic pH a larger proportion is present in the neutral, membrane- permeant form.
In psychotic excitement (agitation), sedation can be achieved with scopolamine. Unlike atropine, scopolamine exerts a calming and amnesiogenic action that can be used to advantage in anesthetic premedication.
Symptomatic treatment in parkinsonism for the purpose of restoring a dopaminergic-cholinergic balance in the corpus striatum. Antiparkinsonian agents, such as benzatropine, readily penetrate the blood-brain barrier. At centrally equi-effective dosage, their peripheral effects are less marked than are those of atropine.
Contraindications for Parasympatholytics (Anticholinergics)
Glaucoma: Since drainage of aqueous humor is impeded during relaxation of the pupillary sphincter, intraocular pressure rises.
Prostatic hypertrophy with impaired micturition: loss of parasympathetic control of the detrusor muscle exacerbates difficulties in voiding urine.
Atropine Poisoning
Parasympatholytics have a wide therapeutic margin. Rarely life-threatening, poisoning with atropine is characterized by the following peripheral and central effects:
Peripheral Effects: tachycardia; dry mouth; hyperthermia secondary to the inhibition of sweating.
Although sweat glands are innervated by sympathetic fibers, these are cholinergic in nature. When sweat secretion is inhibited, the body loses the ability to dissipate metabolic heat by evaporation of sweat. There is a compensatory vasodilation in the skin allowing increased heat exchange through increased cutaneous blood flow. Decreased peristaltic activity of the intestines leads to constipation.
Central Effects: Motor restlessness, progressing to maniacal agitation, psychic disturbances, disorientation, and hallucinations.
Elderly subjects are more sensitive to such central effects. In this context, the diversity of drugs producing atropine-like side effects should be borne in mind: e.g., tricyclic antidepressants, neuroleptics, antihistamines, antiarrhythmics, antiparkinsonian agents.
Treatment of Atropine Poisoning
Apart from symptomatic, general measures (gastric lavage, cooling with ice water), therapy of severe atropine intoxication includes the administration of the indirect parasympathomimetic physostigmine.
The most common instances of “atropine” intoxication are observed after ingestion of the berry-like fruits of belladonna (children) or intentional overdosage with tricyclic antidepressants in attempted suicide.