Table of Contents
I. Replacement therapy
The adrenal cortex (AC) produces the glucocorticoid cortisol (hydrocortisone) and the mineralocorticoid aldosterone. Both steroid hormones are vitally important in adaptation responses to stress situations, such as disease, trauma, or surgery.
Cortisol secretion is stimulated by hypophyseal ACTH, aldosterone secretion by angiotensin II in particular. In AC failure (primary AC insuffiency: Addison’s disease), both cortisol and aldosterone must be replaced; when ACTH production is deficient (secondary AC insufficiency), cortisol alone needs to be replaced.
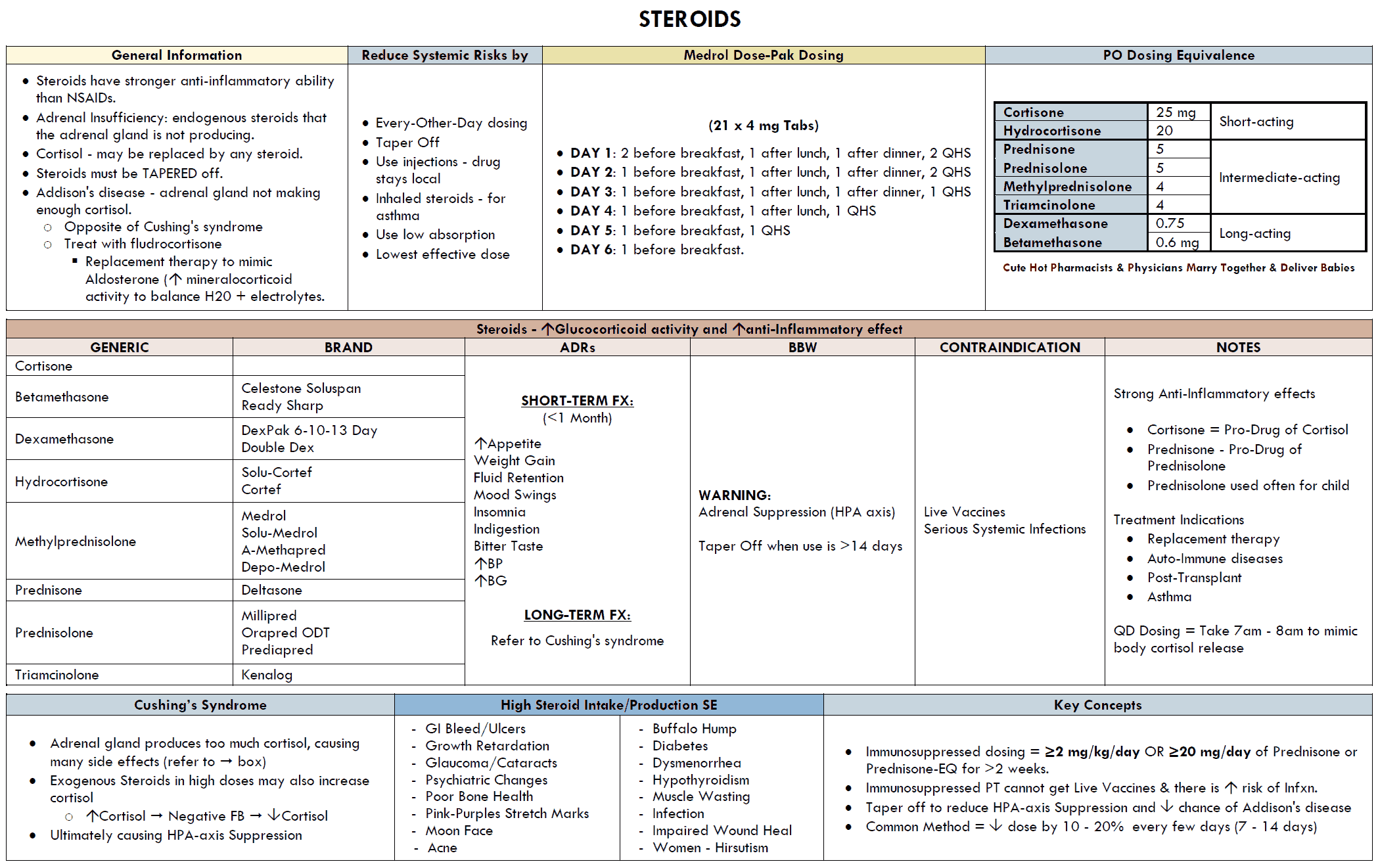
Cortisol is effective when given orally (30 mg/d, 2/3 a.m., 1/3 p.m.). In stress situations, the dose is raised by 5- to 10-fold. Aldosterone is poorly effective via the oral route; instead, the mineralocorticoid fludrocortisone (0.1 mg/d) is given.
II. Pharmacodynamic therapy with glucocorticoids
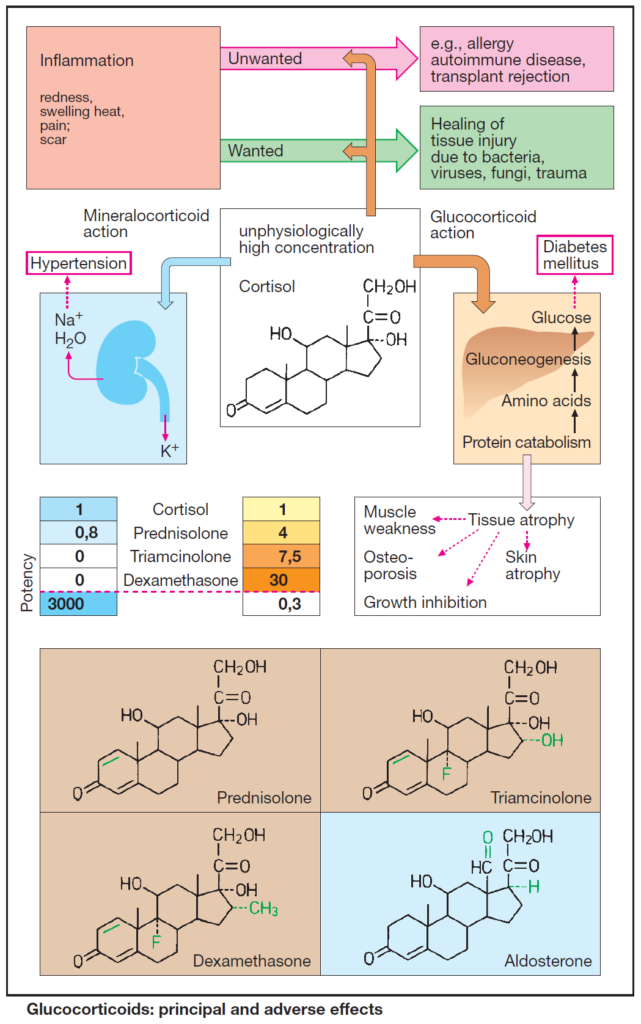
In unphysiologically high concentrations, cortisol or other glucocorticoids suppress all phases (exudation, proliferation, scar formation) of the inflammatory reaction, i.e., the organism’s defensive measures against foreign or noxious matter.
This effect is mediated by multiple components, all of which involve alterations in gene transcription. Glucocorticoids inhibit the expression of genes encoding for proinflammatory proteins (phospholipase-A2, cyclooxygenase 2, IL-2-receptor).
The expression of these genes is stimulated by the transcription factor NFΚB. Binding to the glucocorticoid receptor complex prevents translocation af NFΚB to the nucleus. Conversely, glucocorticoids augment the expression of some anti-inflammatory proteins, e.g., lipocortin, which in turn inhibits phospholipase A2.
Consequently, release of arachidonic acid is diminished, as is the formation of inflammatory mediators of the prostaglandin and leukotriene series. At very high dosage, nongenomic effects may also contribute.
Desired effects
- anti-allergics
- immunosuppressants
- anti-inflammatory
Unwanted effects
With short-term use, glucocorticoids are practically free of adverse effects, even at the highest dosage.
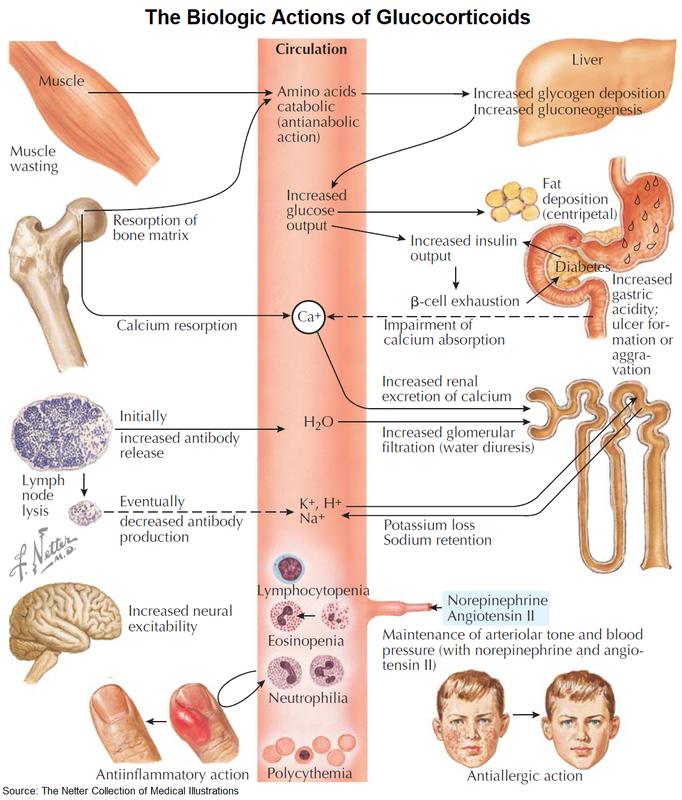
Long-term use is likely to cause changes mimicking the signs of Cushing’s syndrome (endogenous overproduction of cortisol).
Sequelae of the anti-inflammatory action:
- lowered resistance to infection
- delayed wound healing
- impaired healing of peptic ulcers
Sequelae of exaggerated glucocorticoid action:
- increased gluconeogenesis and release of glucose
- insulin-dependent conversion of glucose to triglycerides (adiposity mainly noticeable in the face, neck, and trunk)
- “steroid-diabetes” if insulin release is insufficient
- increased protein catabolism with atrophy of skeletal musculature (thin extremities)
- osteoporosis
- growth retardation in infants
- skin atrophy
Sequelae of the intrinsically weak, but now manifest, mineralocorticoid action of cortisol:
- salt and fluid retention
- hypertension
- edema
- KCl loss with danger of hypokalemia